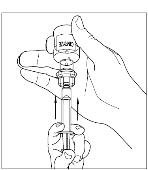
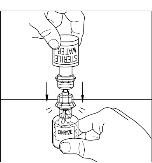
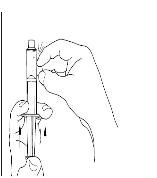
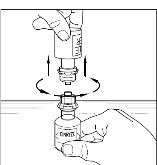
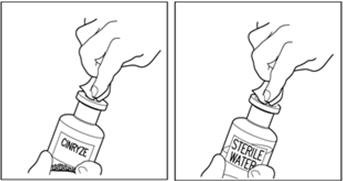
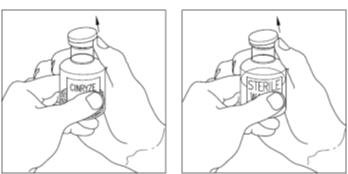
Final Results from the Longest Hereditary Angioedema Study of Active Treatment Duration Conducted to Date Support the Sustained Safety and Efficacy of TAKHZYRO® (lanadelumab) Injection for Long-Term Prevention of Hereditary Angioedema Attacks

Recombinant replacement therapy for hereditary angioedema due to C1 inhibitor deficiency | Immunotherapy

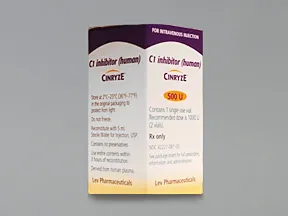
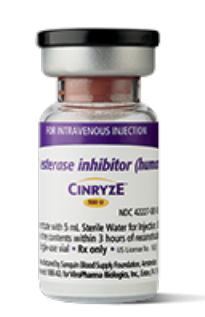
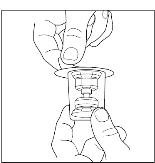
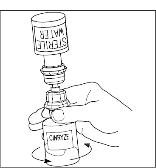
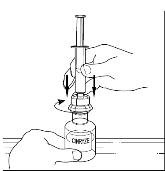
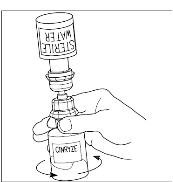
Takeda launches CINRYZE™ in India, the first C1-I NH for prophylaxis in hereditary angioedema patients